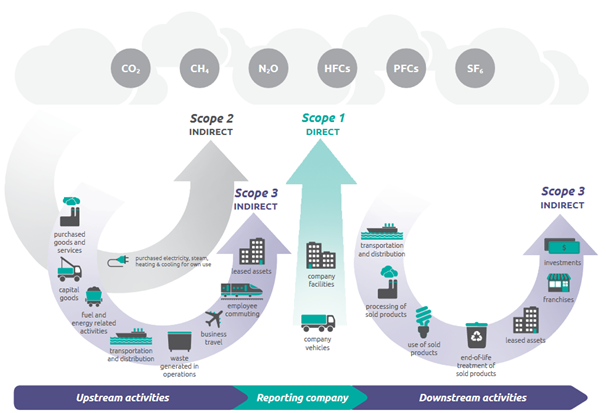
Lab activity 7 post lab - d. If the water vapor pressure was not subtracted from the atmospheric - Studocu

JRC Publications Repository - Methodological overview on the calculation of air pollutant and greenhouse gas emissions from agricultural activities

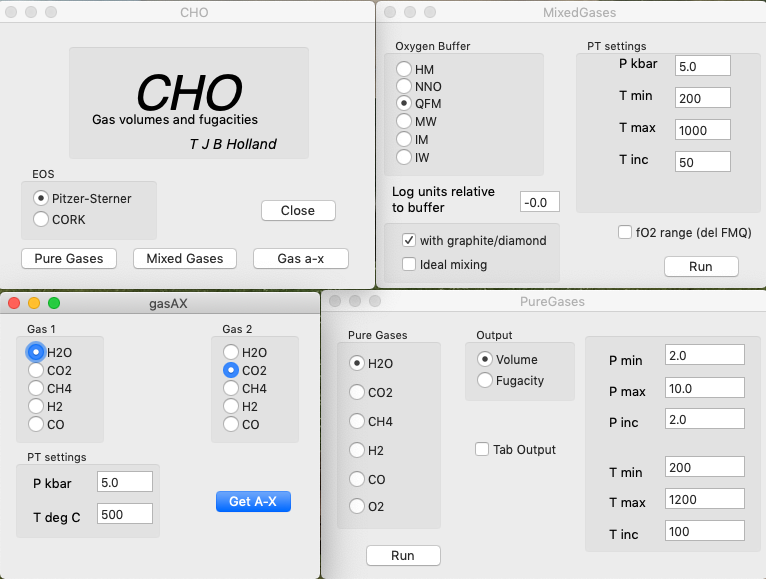
HSC Chemistry, Software for Process simulation, Reactions Equations, Heat and Material Balances, Heat Loss Calculator, Equilibrium Calculations, Electrochemical Cell Equilibriums, Eh-pH Diagrams – Pourbaix diagram, Tpp Diagrams – Stability diagrams ...

SOLVED: Question L a) At T= 100 K, 1.00 mol of COz occupies volume of 1.00 L. Calculate the pressures given by the ideal gas equation and the van der Waals equation: (
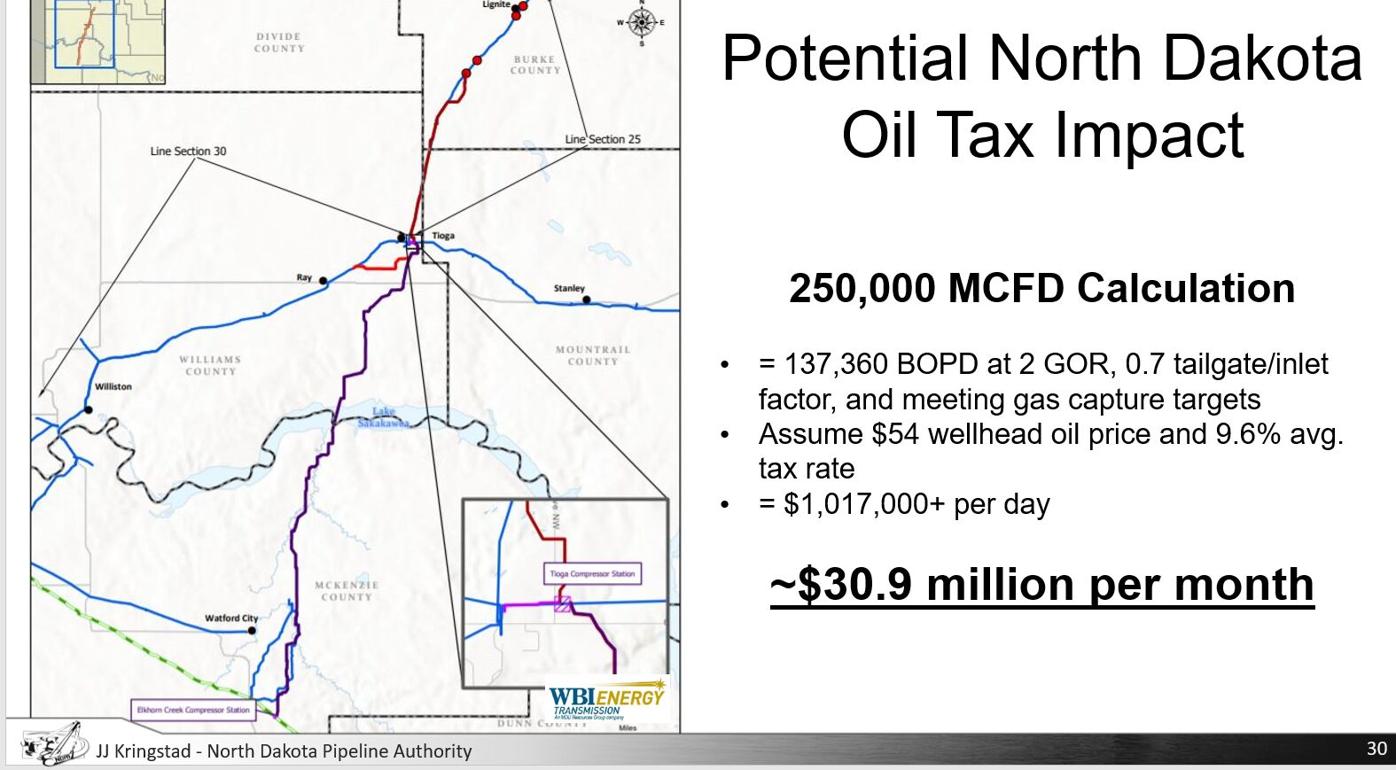
Oil and gas activity north of Lake Sakakawea is stronger, following completion of WBI's North Bakken Expansion | Oil And Energy | willistonherald.com

SOLVED: 'A sealed flask contains the following substances: 75 L of 0.00042 M NazSO4(aq) 21g sand (SiOz(s)) 0.5 bar Nz(g) 0.10 bar Ar(g) Calculate the activity of each substance in the flask
![PDF] Activity Based Costing Model for Cost Calculation In Gas Companies: Empirical Evidence of Iran | Semantic Scholar PDF] Activity Based Costing Model for Cost Calculation In Gas Companies: Empirical Evidence of Iran | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/389ee595c3577b118470791cd43a4af9838e2ef7/4-Table2-1.png)
PDF] Activity Based Costing Model for Cost Calculation In Gas Companies: Empirical Evidence of Iran | Semantic Scholar