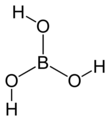
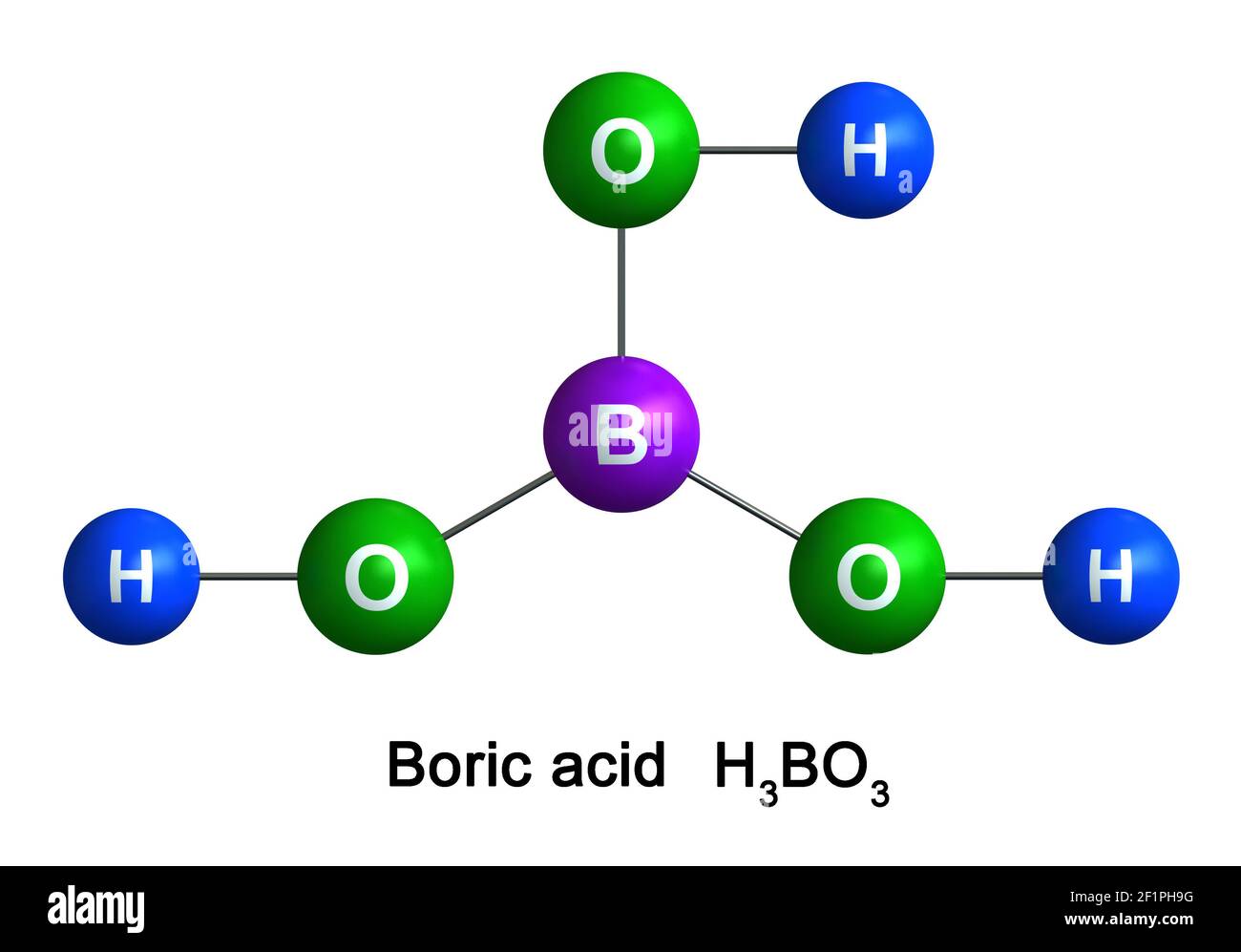
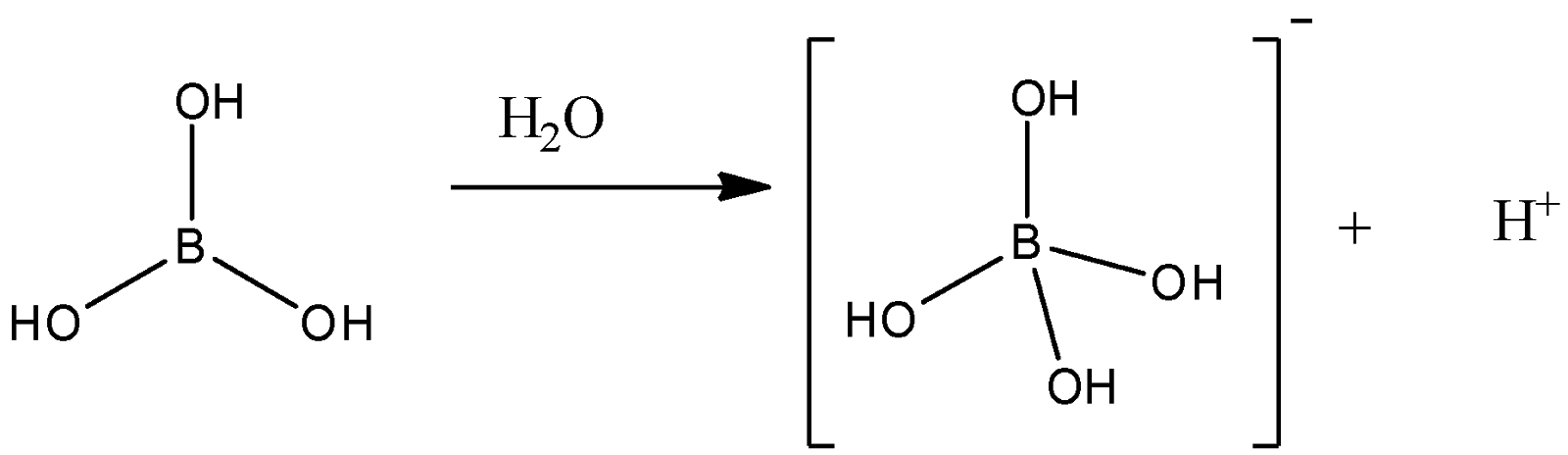
Welcome to Chem Zipper.com......: Which of the following correct statement about structure of Ortho boric acid (H3BO3 ) is/are? Statements are as

B2O3 + 3 H2O ---> 2 H3BO3 If diboron trioxide is reacted with water, the product is boric acid. What mass of boric acid is obtained from the full reaction. - ppt video online download

Orthoboric acid, on heating, decomposes in two ways: (I) H3BO3 → HBO2 + H2O (II) H5BO3 → B2O3 + H2O If 9 moles of H3BO3 decomposes by (I) pathway and remaining by (

![Boric Acid [H3BO3] Molecular Weight Calculation - Laboratory Notes Boric Acid [H3BO3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/boric-acid-molecular-weight-calculation-300x208.jpg)